
Practice · Pharmaceutical + life sciences
Regulated utility problems, held to inspection standard.
ASE started in pharmaceutical water in 2003. Every discipline the firm has added since — power continuity, controls visibility, private-office infrastructure, and operating software — carries the documentation standard we learned in regulated manufacturing. Pharma is still the benchmark we hold the rest to.
- Practice founded
- 2003
- Water grade
- WFI
- Automation
- GAMP 5
- Operating offices
- 6
Representative delivery posture. Validation depth and retained coverage are set per mandate in the written brief.
Delivered against
- USP <1231>
- EP 0169
- GAMP 5
- ICH Q9
- WHO GMP
Capabilities
Six pharmaceutical programmes under one partner.
Each programme carries the documentation a regulator would accept. The operator, the auditor, and the partner read from the same record.

01
Purified + WFI water
Generation, storage, and distribution loops engineered to USP and EP specification. Hot-loop sanitisation, full validation packs, and qualification support.

02
Clean-utility generation
Clean steam, compressed air, and process gases specified against the actual point-of-use demand — not the vendor package.
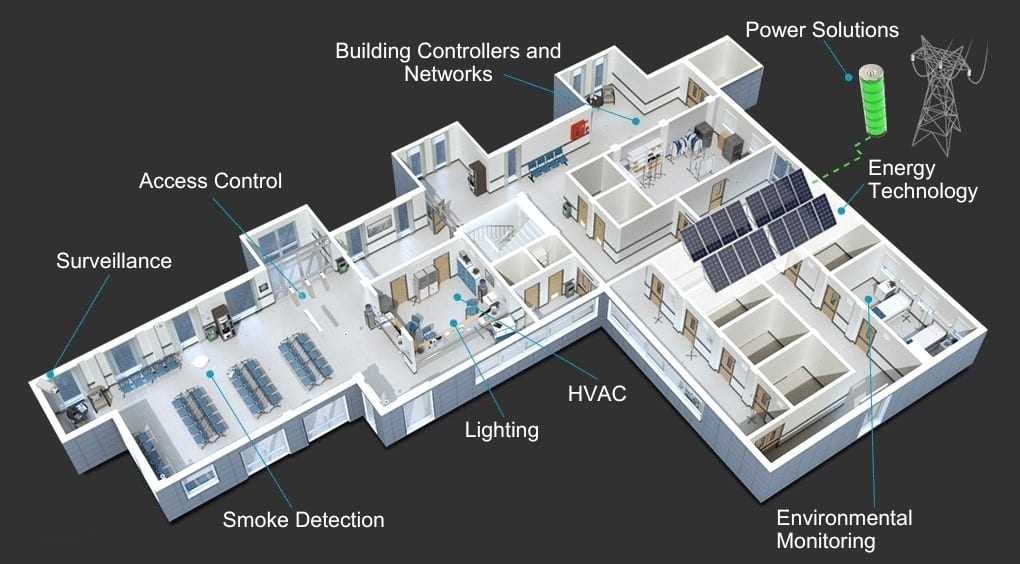
03
GAMP-aligned automation
PLC, SCADA, and historian systems delivered with control narrative, role matrix, and audit trail acceptable to regulators.

04
Facility continuity
Redundancy engineering for HVAC, electrical, and critical utilities. Written impact analysis before any single-point-of-failure is accepted.

05
CSV + validation lifecycle
IQ, OQ, and PQ delivery, periodic review, and change control run by the engineers who designed the system — not subcontracted to a third party.

06
Remediation + inspection response
CAPA engineering response for regulator findings. Written plan, named owner, and evidenced close-out inside the client’s deadline.

The discipline learned in pharmaceutical water became the documentation standard for every other practice the firm later built.
Evidence
A regulated-industry practice since 2003.
Four declarations drawn from the firm’s own record. Stated in writing before any equipment is specified.
Record of practice
i — iv · as at 2026
- ILineage
2003
Year ASE was founded on pharmaceutical water — the practice the firm grew from
- IIStandard
USP / EP
Compliance regimes delivered end to end with full validation support
- IIIFootprint
6 offices
Vienna · Lisbon · Bucharest · Lagos · Abuja · Accra
- IVPosture
NDA
Default engagement posture for regulated manufacturers and private clients
Signed by the senior engineer of record at the close of every engagement. Kept on file, reissued on request.
Asset class
The pharma work we sign for.
Representative images of the treatment, qualification, and control assets ASE specifies and commissions on regulated sites — the asset class, not specific client facilities.

Facility
Regulated plant room.
Pre-treatment, RO, EDI, and distribution loops commissioned as one documented system, signed for by our lead engineer.

Qualification
Validation on delivery.
IQ, OQ, PQ, and periodic-review packs issued with the system — not bolted on months after handover.

Distribution
Hot-loop sanitisation.
Distribution loops designed against the incoming analysis and the point-of-use profile. No generic brochure train.

Controls
GAMP-ready automation.
Control narrative, role matrix, audit trail, and 21 CFR Part 11-aligned records delivered with the system.
Delivery
Survey. Design. Build + qualify. Steward.
The discipline that regulators expect inside the facility applies to the delivery record outside it. One partner signs across all four stages.
Engagement cadence
i — iv · single partner
- IListen
Survey
Site walk, utility demand modelling, and URS review. A written baseline of the facility before any specification is drafted.
1 – 2 weeks
- IIWrite
Design
P&IDs, equipment selection, and validation strategy issued as a stamped package. Independent of any single equipment vendor.
3 – 6 weeks
- IIIDeliver
Build + qualify
Installation, FAT, SAT, sanitisation, and IQ/OQ delivery under the name of the partner who signed the scope.
12 – 24 weeks
- IVHold
Stewardship
Periodic review, change control, and planned replacements by the engineers who designed the system.
Retained
One partner signs across all four stages. No subcontracted validation, no handover across firms.
More on pharmaceutical water and regulated-utility delivery in the ASE journal.
Next step
A partner-level facility review.
Thirty minutes with a senior partner from the pharma practice. You leave with a written summary, a realistic view of the work, and a direct line back to the partner.